- Blog
- Hyperspin game themes download
- Kofax vrs no such resource
- Game face ufc undisputed 3
- Diljale 1996 hindilink4u-to
- How to calculate ppm from -
- Aams mastering keygen
- Centos 7 static ip address configuration
- Octopath traveler ost download free mp3
- Forza motorsport 4 tune setups
- Stop shutdown timer windows 10
- Illusion games discord
- Imvu badges gratis
- Tamil god songs free download for mobile
- X4500mhd benchmark
- Bonnie bedilla judicial consent sex scenes
Although rarely used in science (ppm is typically used instead), one permyriad has an unambiguous value of one part per 10,000 (10 4) parts, and a value of 10 −4.
How to calculate ppm from % full#
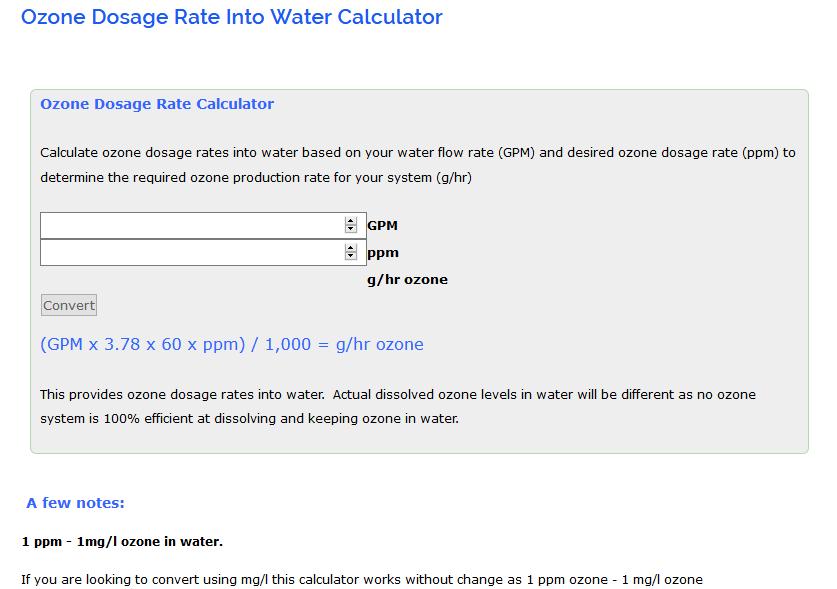
Consequently, 1 ppm corresponds to 1 mg/L and 1 ppb corresponds to 1 μg/L. Therefore, it is common to equate 1 kilogram of water with 1 L of water. When working with aqueous solutions, it is common to assume that the density of water is 1.00 g/mL. The quantity "1 ppm" can be used for a mass fraction if a water-borne pollutant is present at one-millionth of a gram per gram of sample solution. Parts-per notation is often used describing dilute solutions in chemistry, for instance, the relative abundance of dissolved minerals or pollutants in water.
